Medical Device Design Services
ISO 13485 Certified
Comprehensive Medical Device Design & Consulting Services
End-to-End Medical Device Development
As a leading medical device design company, Synectic provides end-to-end medical device design services to bring innovative healthcare solutions to market. With decades of experience, our team of expert medical device consultants specializes in developing safe, effective, and manufacturable devices that meet stringent regulatory standards.
From concept to commercialization, we ensure a seamless development process, helping startups and established companies navigate complex regulatory landscapes while optimizing designs for manufacturability and user experience.
We offer medical device design services across various disciplines, tailoring development to the specific challenges of each area. Our team has experience creating cutting-edge devices that enhance patient care, improve clinical outcomes, and meet the highest regulatory standards in the following medical disciplines:
Cardiology • Orthopedics • Obstetrics & Gynecology • Pediatrics • Dentistry • Gastroenterology • General Surgery • Diagnostics & Radiology • Physical Medicine & Rehabilitation • Endocrinology • Ophthalmology • Hematology • Nephrology • Geriatrics
Expertise Across Healthcare Markets
Handheld Surgical Devices
Implantable Devices
Wearable & Connected Devices
Drug Delivery Systems
Robotic Surgery Systems
Diagnostic & Imaging Equipment
Our Medical Device Design Services
Design & Development
Clinician Feedback
VOC Studies
In-Vitro & In-Vivo Testing
Design Verification & Validation
Cleanroom Assembly
Human Factors Engineering
Biocompatibility Testing
Packaging Design & Testing
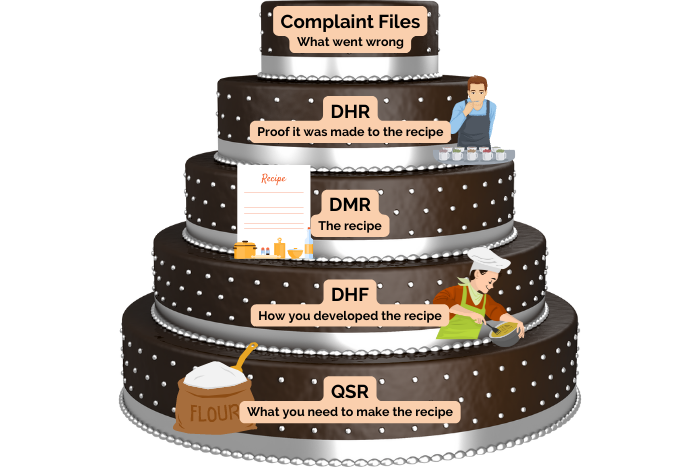
Quality Planning & Management
21 CFR Design Controls
Sterilization Testing
DHF Management
Feasibility Studies
Risk Assessment
Design Transfer
Why Partner with Synectic?
- Deep Industry Knowledge – Our medical device consultants have extensive experience designing Class I, II, and III devices.
- User-Centered Approach – We prioritize patient safety and usability in every design.
- Seamless Path to Manufacturing – Our integrated approach within the Mack Group ensures smooth transitions from design to production. We not only get your device to market faster but also eliminate costly manufacturing and development do-loops.
- Regulatory Excellence – We stay ahead of evolving compliance requirements to streamline approvals with our FDA-focused proprietary quality documentation system.
- Collaborative Innovation – We work closely with your team to refine ideas and accelerate development timelines.
- Proven Track Record – Our portfolio includes dozens of successful projects spanning a wide range of medical disciplines.