MOST POPULAR WHITE PAPERS
ALL WHITE PAPERS

The Hidden Cost of Inventing: Fixed-Fee vs. Time-and-Materials
In the world of product development, inventors looking to keep costs controlled are leaning more and more towards outsourcing engineering services to a product development company. As with any client/vendor ... Read More
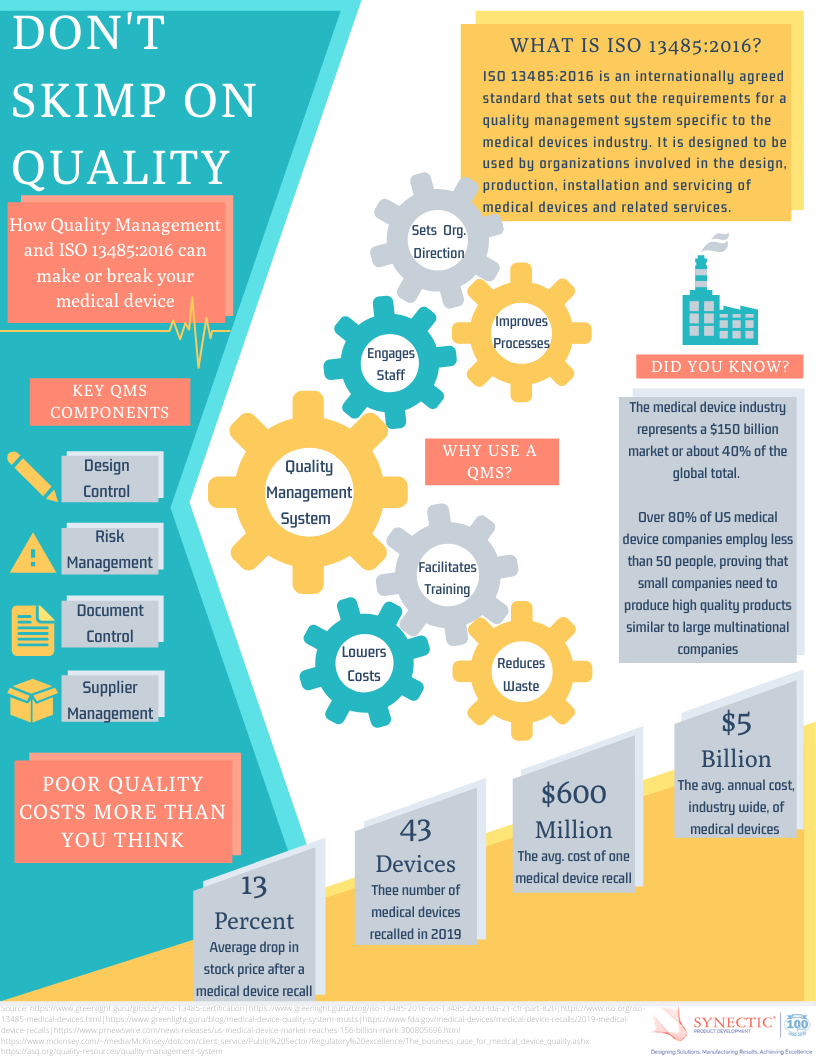
Quality Management Systems and ISO 13485:2016 Infographic
Need help developing an ISO compliant QMS? Speak with one of our experts and get answers to all your project questions. Request a Quote About Synectic Product Development: Synectic Product ... Read More

How to Invent Something: 3 Essentials to Get Started
Inventing in today's ever-changing business market can be daunting. Inventors need to establish a business plan, raise funds, and find a capable product design company before product development can begin ... Read More

New Product Introduction Guide – How the NPI Process Works
If you are asking how to launch your product, you must first ask yourself if your product is ready for manufacturing and market launch. If you respond, “Of course I am ... Read More

How to Get a Patent
Do you have an idea but are uncertain how to patent that idea? You are not alone. Navigating the world of patents is not easy and can be incredibly frustrating ... Read More

What is Machining? A Machining FAQ
Machining, also known as subtractive manufacturing, is a prototyping and manufacturing process that creates the desired shape by removing unwanted material from a larger piece of material. What are the different types of machining? ... Read More

What is Rapid Prototyping? A Rapid Prototyping FAQ
Rapid prototyping, also known as additive manufacturing, is a prototyping method that makes three-dimensional physical objects from a digital file. A 3D printer lays down many layers of material in succession, building ... Read More
The Product Development Process
The product development process takes an inventor step-by-step from a concept to production. Every company may call the phases by different names or include slightly different deliverables in each phase, ... Read More






